Ankle Replacement
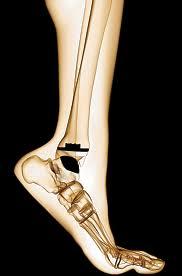 Ankle Replacement
Ankle Replacement
Many Americans are aware of the options available to orthopedic patients suffering from severe or dysfunction of hip and knee replacement surgeries, but awareness of ankle replacement procedures is less common. Ankle replacement procedures, medically known as 'arthroplasty', though much less common than hip or knee replacements, have been increasing in numbers in the United States in recent years. With the baby-boomer population growing older, more Americans are opting for ankle replacement procedures for painful conditions such as osteoarthritis as these procedures can relieve pain, stabilize joints, and lead to greater range of motion. The term 'arthroplasty' means 'surgical repair of the joint,' as this procedure repairs or replaces joint components. According to the American College of Foot and Ankle Surgeons (ACFAS), ankle replacements have more than doubled in the U.S. in recent years, partly due to technological advances in ankle prosthetics.
History of ankle replacement surgery
In 1893, Dr. Jules Pean performed the first successful arthroplasty in the United States. In the 1950s, Dr. Stephen Hudack from New York City tested artificial joints on animals and soon began replacing human hip joints. In the past, fusion or arthrodesis were used in the treatment severe pain caused by such medical conditions as rheumatoid arthritis, osteoarthritis, or trauma involving ankles. Unfortunately, procedures such as fusion, though effective for pain relief, also rendered the ankle immobile. By this time, the method of using artificial materials for joint replacement became an accepted method in several countries around the world for relieving severe joint pain due to trauma or medical conditions such as arthritis. Ankle arthroplasties are rapidly on the rise as these surgeries involve implanting medical devices to relieve pain and restore range of motion. Currently, patients receiving these types of procedures are typically in the 40s to 60s, but older patients who are active and in good health can receive them in order to continue leading active lives.
About ankle replacement implants
The AAOS reports that early ankle replacement designs had only two components and required bone cement to hold them in place. Newer medical devices for ankle arthroplasty do not require cement, but instead bone grows into the ankle implant. Although total ankle replacement procedures have been around for more than a quarter century, recent breakthroughs have increased options regarding effective implants. In the late 20th century, only one ankle prosthesis was approved by the FDA, whereas today in the U.S., there are four FDA-approved ankle implants on the market. To date, the FDA has approved five ankle prostheses, while several others are being developed and researched nationwide. Just as with other joint replacement surgeries, some of the different types of implant components used in ankle arthroplasty today are metal and plastic, titanium, cobalt, and polyethylene.
Pharmaceutical regulations and compliance
Ankle replacement implants have not had the negative press recently in the United States as some hip replacement devices have, largely due to the metal-on-metal components used in these hip arthroplasties. To date, the Food and Drug Administration (FDA) has approved four 2-component ankle prosthetics for use in the U.S. (In other countries, orthopedic surgeons have been using 3-component ankle devices for years, as 3-components implants reportedly allow for more range of motion in the joint.) The FDA recently approved a total ankle replacement system for patients with deformed or severely arthritic ankles. The new implant is a mobile-bearing device, meaning it relies on bearings that move across a polyethylene surface. This surface made of polyethylene is favorable because it is a flexible plastic. This new device approved by the FDA is the first of its kind. As with all medical devices, including all implants for other types of replacement procedures such as for hip, knee, and shoulder replacements, the FDA recently began enforcing stricter regulations for all manufacturers of arthroplasty prostheses. Manufacturers looking to sell ankle implants must file applications that include not only results of clinical trials, but also scientific evidence proving that their ankle replacement devices are both safe and effective for use in ankle arthroplasties. Any manufacturers who do not meet these stricter standards and are unable to prove the safety of their devices will be denied the ability to sell them in the United States.
History of ankle replacement surgery
In 1893, Dr. Jules Pean performed the first successful arthroplasty in the United States. In the 1950s, Dr. Stephen Hudack from New York City tested artificial joints on animals and soon began replacing human hip joints. In the past, fusion or arthrodesis were used in the treatment severe pain caused by such medical conditions as rheumatoid arthritis, osteoarthritis, or trauma involving ankles. Unfortunately, procedures such as fusion, though effective for pain relief, also rendered the ankle immobile. By this time, the method of using artificial materials for joint replacement became an accepted method in several countries around the world for relieving severe joint pain due to trauma or medical conditions such as arthritis. Ankle arthroplasties are rapidly on the rise as these surgeries involve implanting medical devices to relieve pain and restore range of motion. Currently, patients receiving these types of procedures are typically in the 40s to 60s, but older patients who are active and in good health can receive them in order to continue leading active lives.
About ankle replacement implants
The AAOS reports that early ankle replacement designs had only two components and required bone cement to hold them in place. Newer medical devices for ankle arthroplasty do not require cement, but instead bone grows into the ankle implant. Although total ankle replacement procedures have been around for more than a quarter century, recent breakthroughs have increased options regarding effective implants. In the late 20th century, only one ankle prosthesis was approved by the FDA, whereas today in the U.S., there are four FDA-approved ankle implants on the market. To date, the FDA has approved five ankle prostheses, while several others are being developed and researched nationwide. Just as with other joint replacement surgeries, some of the different types of implant components used in ankle arthroplasty today are metal and plastic, titanium, cobalt, and polyethylene.
Pharmaceutical regulations and compliance
Ankle replacement implants have not had the negative press recently in the United States as some hip replacement devices have, largely due to the metal-on-metal components used in these hip arthroplasties. To date, the Food and Drug Administration (FDA) has approved four 2-component ankle prosthetics for use in the U.S. (In other countries, orthopedic surgeons have been using 3-component ankle devices for years, as 3-components implants reportedly allow for more range of motion in the joint.) The FDA recently approved a total ankle replacement system for patients with deformed or severely arthritic ankles. The new implant is a mobile-bearing device, meaning it relies on bearings that move across a polyethylene surface. This surface made of polyethylene is favorable because it is a flexible plastic. This new device approved by the FDA is the first of its kind. As with all medical devices, including all implants for other types of replacement procedures such as for hip, knee, and shoulder replacements, the FDA recently began enforcing stricter regulations for all manufacturers of arthroplasty prostheses. Manufacturers looking to sell ankle implants must file applications that include not only results of clinical trials, but also scientific evidence proving that their ankle replacement devices are both safe and effective for use in ankle arthroplasties. Any manufacturers who do not meet these stricter standards and are unable to prove the safety of their devices will be denied the ability to sell them in the United States.
